RealPeopleGroup/E+ through Getty Pictures
Biotech investing is filled with surprises, generally as a result of analysts have an unsophisticated view as to how difficult doing new stuff may be. Seres (NASDAQ:MCRB) has simply obtained FDA approval for the primary oral (pill) remedy for stopping recurrence of a significant hospital (and aged care facility) bacterial an infection. Right here I have a look at the Seres share value fall in response to FDA approval. It is unnecessary to me.
Monitoring Seres share value
Seres 5 12 months share value chart (In search of Alpha)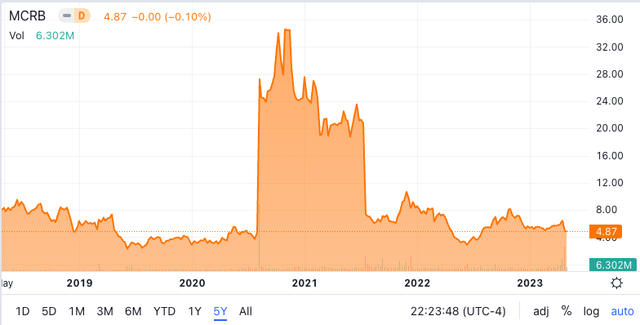
The above 5-year share value chart for Seres exhibits two dramatic occasions that strongly influenced the share value.
The primary was in August 2020 when, after an unsuccessful Part 2 trial in 2016 which brought about the share value to fall beneath $10, SER-109 lastly achieved success with its Ecospor III Part 3 trial. This dramatic share value enhance from ~$4 to ~$25 was adopted by additional share price rise to $32 on additional bulletins regarding success of the Part 3 examine for SER-109 in October 2020.
The second dramatic occasion was in July 2021, this time a dramatic fall in share value from $21 to beneath $7 on the failure of Seres’ Part 2 trial regarding its microbiomic remedy for Ulcerative Colitis (UC). Plenty of commentary on this consequence represented pessimism about Seres’ potential to develop a microbiomic remedy for this large unsolved inflammatory bowel situation. The commentary neglected the truth that Seres failed beforehand in its improvement of SER-109 to deal with C.diff recurrence, earlier than in the end succeeding. The jury remains to be out on Seres UC microbiomic drug strategy, however Seres signifies that it continues that program.
The above two vital occasions are key to the present Seres share value. It has been caught between $5 and $10 since mid-2021.
Why has the share value gone down with FDA approval?
There have been 6.18 million Seres shares traded on the day of the FDA approval and the share value completed at $6.40. The day after FDA approval 20.76 million shares had been traded and the share value ended at $5.30. Total within the 4 days since approval, 41.83 million shares have been traded, with the share value falling to $4.67 on quantity of 4.7 million on Tuesday. The share value could also be stabilising at roughly $1 above its buying and selling value earlier than the profitable Part 3 trial for SER-109 that put the corporate on a trajectory for FDA approval of SER-109.
I’ve seen some commentary that appears to assume that Seres is caught in the beginning of the microbiomic revolution with a product that’s derived from processed fecal samples at a time when others are exploring extra outlined merchandise comprising cultured micro organism. Because of this it appears the logic is to disregard Seres as a result of higher investments are coming, in the event you settle for that microbiomics goes to turn out to be a big a part of the biotech remedy profile. This can be a maybe an excessive instance of purchase on hope and promote on actuality. The purpose is that Seres has an preliminary fecal-derived product that has had two very profitable Part 3 trials for treating recurrence of essentially the most important bacterial an infection in hospitals (and aged care services) and the product might be prepared on the market in June (lower than 30 days). The analyst view appears to be to disregard the advance and upcoming income as a result of a while sooner or later there might be higher options.
I believe the above view wants assist from technical consultants who know concerning the difficulties of culturing anaerobic micro organism and certainly culturing organisms which can be solely recognized from their DNA sequence. Then there’s the problem of which micro organism to place within the ultimate product. That is about as near rocket science that biotech will get. Seres is a frontrunner on this space and it’s already evaluating two outlined merchandise comprising cultured micro organism (i.e. not from processed fecal materials).
Maybe these with a adverse view of Seres could not even bear in mind that Seres is a frontrunner in defining microbiomic merchandise primarily based cultured micro organism? Seres has SER-155 in Part 1b scientific trial for complicated bacterial infections. SER-155 has been designed primarily based on findings from their SER-109 program.
A second cultured product SER-301 has a extra complicated historical past. It’s a outlined cultured group of micro organism developed from the fecal-derived SER-287. Ulcerative Colitis is a large alternative, but additionally a really robust problem. At present Seres is reviewing its biomarker information from SER-287 and SER-301 to discover the following steps of its UC program. Growth in uncharted territory isn’t a straight line.
So far as I’m conscious, no firm has an outlined product in Part 3 trials but. Which means that validation of scientific effectiveness is a good distance from being realised. This contains the outlined merchandise from the Seres portfolio.
After all I could also be lacking the way in which the market thinks and the falling share value could also be all a few attainable “miss” within the upcoming Q1 earnings expectations, which in my opinion has nearly no relevance to the corporate’s future efficiency.
My tackle the present Seres share value
The fascinating factor is that, however the continued progress with the profitable VOWST (SER-109) C.diff program, the share value has been caught beneath $10 since mid-2021. A profitable Part 3 trial regarding SER-109 took the share value as much as $32 and a subsequent failed early (Part 2) trial with SER-287 on UC introduced the share value beneath $10. Since then Seres (and Nestle (OTCPK:NSRGY)) have progressed the SER-109 program with a second profitable Part 3 trial, however the share value has remained largely ~$6. Presumably the market remained skeptical about Seres potential to attain FDA approval as a result of plenty of different microbiomic corporations have faltered in packages transferring in the direction of FDA approval.
Due to the above I assumed that reaching FDA approval for its pill type of SER-109, an enormous milestone for any microbiomic product, would result in a share value enhance, noting that the preliminary profitable Part 3 trial led to a $32 share value spike.
As an alternative the share value fell beneath $5 on the latest FDA approval to provide Seres an enterprise worth of simply $604 million. That is very odd, particularly when Seres and Nestle have introduced that VOWST (previously SER-109) might be out there for sufferers in June 2023!! Clearly Seres and Nestle took optimistic alerts from the FDA earlier this 12 months to point that approval would come on April 26 (because it did) they usually have been actively concerned with manufacture of VOWST and getting ready for market launch. I counsel June might be time to revisit the Seres share value. Traders with an urge for food for danger may take into consideration the above. After all VOWST needs to be efficient, however the two profitable Part 3 trials and FDA approval are promising alerts.
I’m not a monetary advisor however I’ve lived the biotech business for a very long time. My feedback symbolize my very own conclusions. I hope that they’re of curiosity to you and your monetary advisor as you contemplate the dangerous finish of your funding portfolio. It’s clear to me that biotech is not for everybody!







